Deep expertise in medical device development, built on decades of experience
Medical device development requires more than technical expertise. It calls for a deep understanding of the regulatory environment, documented risk management, and the ability to combine multiple technologies into a single functional and safe solution. We help your company bring new products to market efficiently and in compliance with requirements — without unnecessary redesign cycles.
We have broad experience in a wide range of medical device development projects, with specialist expertise in the design of products involving optomechanics, electromechanics, and injection-moulded parts. Examples of the medical devices we have designed include:
- Imaging devices, such as fundus cameras
- Measurement devices, such as spirometers and skin analysers
- Laboratory equipment
- Surgical implants
- Assistive devices
- Drug delivery devices
- Automation equipment for the pharmaceutical industry
Safe medical devices designed for smooth certification and manufacturing

Risk and quality management as the foundation for certification
In medical device development, risk management is a systematic and documented process. We work within frameworks aligned with standards such as ISO 14971 and ISO 13485, integrating risk and quality management into the development process from the very beginning.
In practice, this means:
- systematic hazard identification and risk analysis
- planning risk control measures as part of the technical design
- documented traceability between requirements, design solutions, and testing
Well-executed risk management reduces product liability risk, supports audit readiness, and helps accelerate the certification process.
Usability is safety — and a regulatory requirement
The intended use and operating environment of medical devices place specific demands on usability and safety. In Europe, the Medical Device Regulation (MDR) requires devices to be designed in a way that minimises the risk of use errors and takes foreseeable misuse into account.
In our design work, we consider:
- clear, error-preventing user interfaces
- ergonomics and the realities of the clinical use environment
- technical solutions that support patient and user safety, such as redundancy, alarms, and protective features
- electrical safety and the related structural requirements
Usability is integrated into risk management to ensure that safety is achieved in practice, not only in specifications.

Designed for manufacturability
A medical device must be both technically precise and capable of being manufactured consistently. Design for Manufacturing (DFM) thinking is embedded already at the concept stage.
This means:
- controlled tolerance chain analysis and identification of critical dimensions
- consideration of manufacturing methods and material choices in the design process
- cost-effective and testable structural design
- support for mass production and production ramp-up
Good manufacturability design reduces unit costs, improves yield, and lowers the risks associated with production ramp-up.
Your partner from concept design to mass production
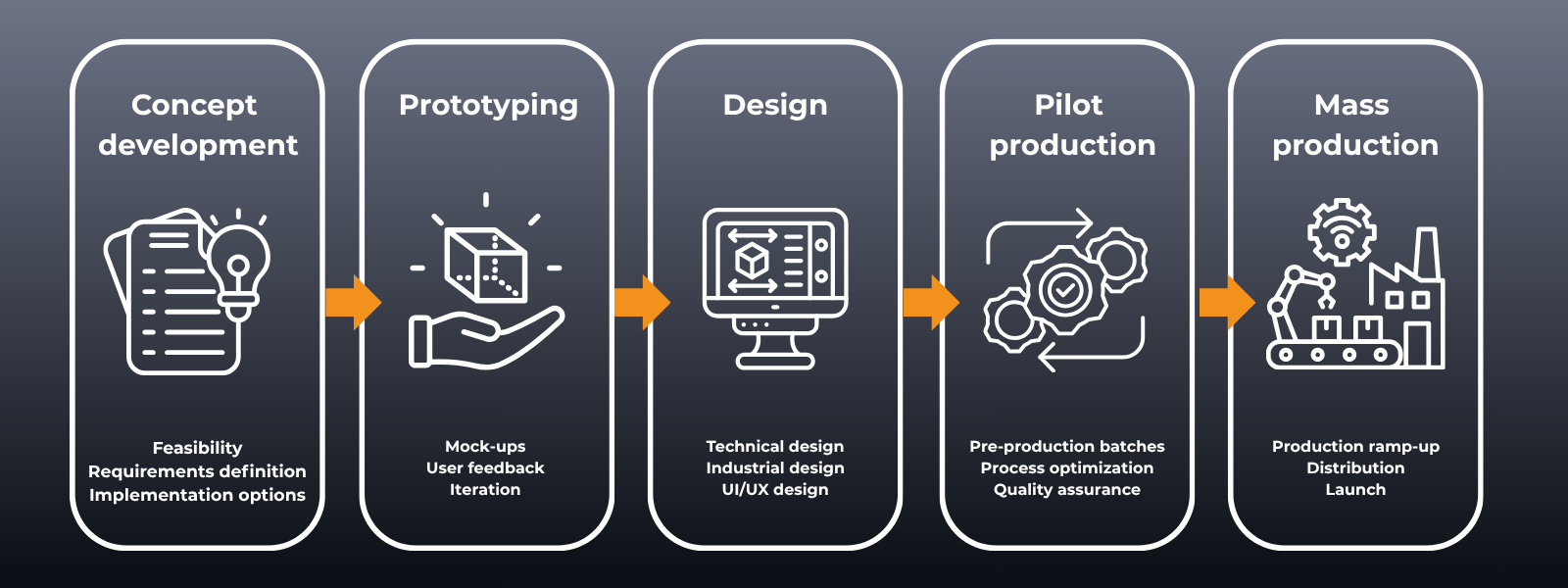
Product Development
We support you at every stage of the product development process, from concept development to mass production. You can entrust the entire development of your product to our experienced team. If you already have an in-house product development team and only need additional resources or specific specialist expertise to complement it, our Rent-a-Designer service offers a flexible solution for expanding your team.
Computer-Aided Engineering
Simulations make it possible to assess product risks and performance already during the design phase, helping to avoid unnecessary prototypes as well as costly and time-consuming iteration cycles. In medical device development, simulations are typically used to analyse factors such as component durability, surface temperatures of enclosure parts, and the heating of sensitive components such as camera sensors and batteries under different operating conditions.
Technical Documentation
From the very beginning of development, we take into account requirements such as MDR as well as the documentation and traceability expected in an ISO 13485 environment. Our expertise in CE marking–related conformity assessment covers everything from the structure of technical documentation to the rationale behind risk management and verification. This helps ensure that the certification and commercialisation phases proceed smoothly.
Tooling And Part Sourcing
Our expertise also covers the sourcing of mass-produced parts and the moulds required for their manufacture. When you outsource mould and component supply to us, we take care of the related administration, mould approvals, incoming inspections, and communication with the factory on your behalf. We work with reliable, high-quality partners for the production of injection-moulded parts and components for medical and optical devices.
What our clients say about us

Optomed Plc pioneered the world’s first handheld fundus camera for the demanding medical device market. From the very beginning, one of the key challenges was to develop highly capable camera optics and retinal illumination, and to package them into a compact, cost-effective product with tight tolerances. Before finding Optotec, we worked with several companies specialising in demanding mechanical design. Once we found Optotec, their expertise proved so valuable that it grew into a long-term and ongoing partnership that has been critical to us. I can warmly recommend Optotec’s broad expertise in demanding mechanical design and automation.
Markku Virta
Chief operating officer at optomed plc (2006-2018)
Are you looking for a partner who understands both the technology and the specific requirements of the medical device environment — and can take your project from concept to market in a controlled and efficient way? Get in touch using the contact form below, by email at sales@optotec.fi, or by phone at +358 40 632 9649.
